 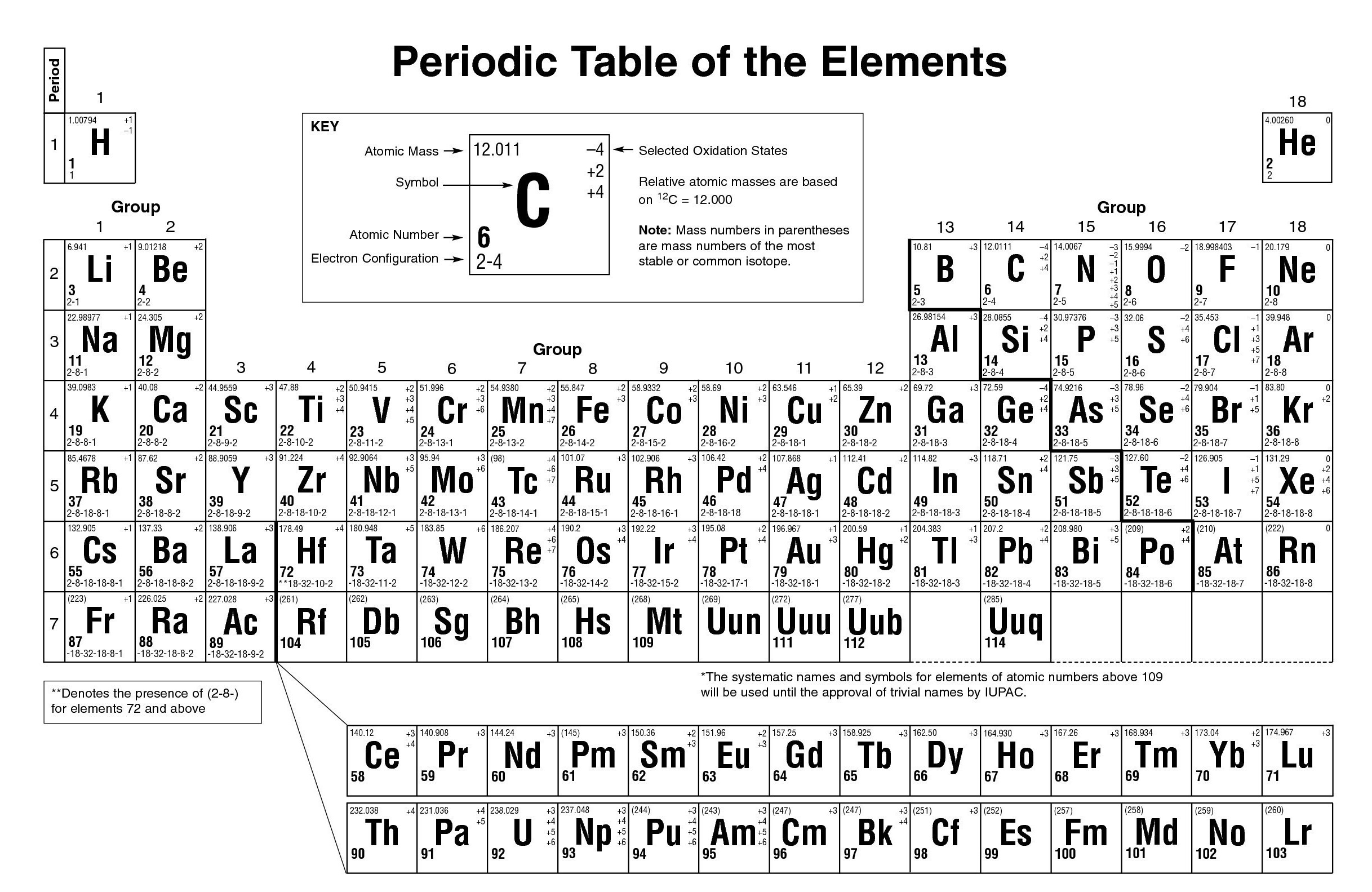
You can effortlessly find every single detail about the elements from this single Interactive Periodic table. Let me tell you how this Interactive Periodic Table will help you in your studies.ġ). You may print and distribute as many paper copies of these periodic tables as you want. You will need to use a nice laser printer because the text is quite small. Free Gift for you: Interactive Periodic Table The following two PDF files can be downloaded and printed on normal 8 1/2 x 11' paper (in landscape orientation) - perfect for including in a 3-ring binder. The hydroxide ions then each have a charge of -1. So, magnesium donates one electron to one hydroxide and one electron to the other hydroxide, giving the Mg atom a +2 charge. Meanwhile, each hydroxide ion gains stability if it gains an electron. In this way, the elements of the same group show similar chemical properties and they also have the same number of valence electrons. In this case, the magnesium ion has two valence electrons in its outer shell. They are soft and can be cut easily with a kitchen knife.Īlso all the elements of group 1 have one valence electron.Īll the elements of group 18 are chemically inert (that means they do not easily react with other elements).Īnd all the elements of group 18 have a complete octet (that means they have 8 electrons in their outer shell). How to calculate charge for ions Find charge using periodic table - Dr K. an ion with a positive charge is called a. The elements lying in the same groups show similar chemical properties and they also have same number of valence electrons.Īll the elements of group 1 are highly reactive to water. IONS: atoms of METALS tend to form positive ions by losing 1 or more e- from their valence (outermost) shell. borate BO3 3- iodate - IO3 sulphite 2- SO3 carbonate 2- CO3 nitrate - NO3 hydrogen sulphide HS. Periodic table with mass is the arrangement of elements in respect to their masses for example hydrogen has a atomic mass of 1.0079. ammonium NH4 + cyanide CN- silicate 2- SiO3 benzoate C6H5COO- hydroxide OH- sulphate SO4 2. There are total 18 vertical columns on periodic table. Group 7 consists of halogens which carry a charge of -1and group 8 consists of noble gases which carry a 0 charge. Groups are the vertical columns on the periodic table. Roman numeral notation indicates charge of ion when element commonly forms more than one ion. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
